Mar 16, 2023 · 7 min read
Here’s Why We’re Opening a New Research Center in Chicago
Priscilla Chan reflects on CZI’s progress in science, which is changing lives and opening extraordinary new horizons in medicine.

CZI will turn eight this year, which is hard to believe. I’ve been thinking a lot about how much we’ve learned since 2015 — and how we’re implementing those learnings as we work together to create impact on a global scale.
In that spirit, I want to dedicate this blog to our progress in science, which is already changing lives — and opening extraordinary new horizons in medicine. I look forward to sharing more areas of our work later in the year.
Unlocking the Mysteries of the Cell
CZ Science has an ambitious mission: to support the science and technology that would make it possible to cure, prevent or manage all diseases by the end of the century.
The first few years of our work were about exploring the ways we can make progress toward this ambitious goal, which led us to our focus for the next 10 years to unlock the mysteries of the cell.
Right now, scientists know very little about how the 37 trillion cells in our bodies develop, specialize and change over time. What we need are ways to measure any biological process within the human body, at any scale, in real time. In that knowledge is the possibility to transform our ability to manage or treat every disease.
That’s precisely what CZI aims to create over the next decade — by building new scientific tools, funding research, and doing awesome science.
Building New Technology To Understand How Our Cells Interact
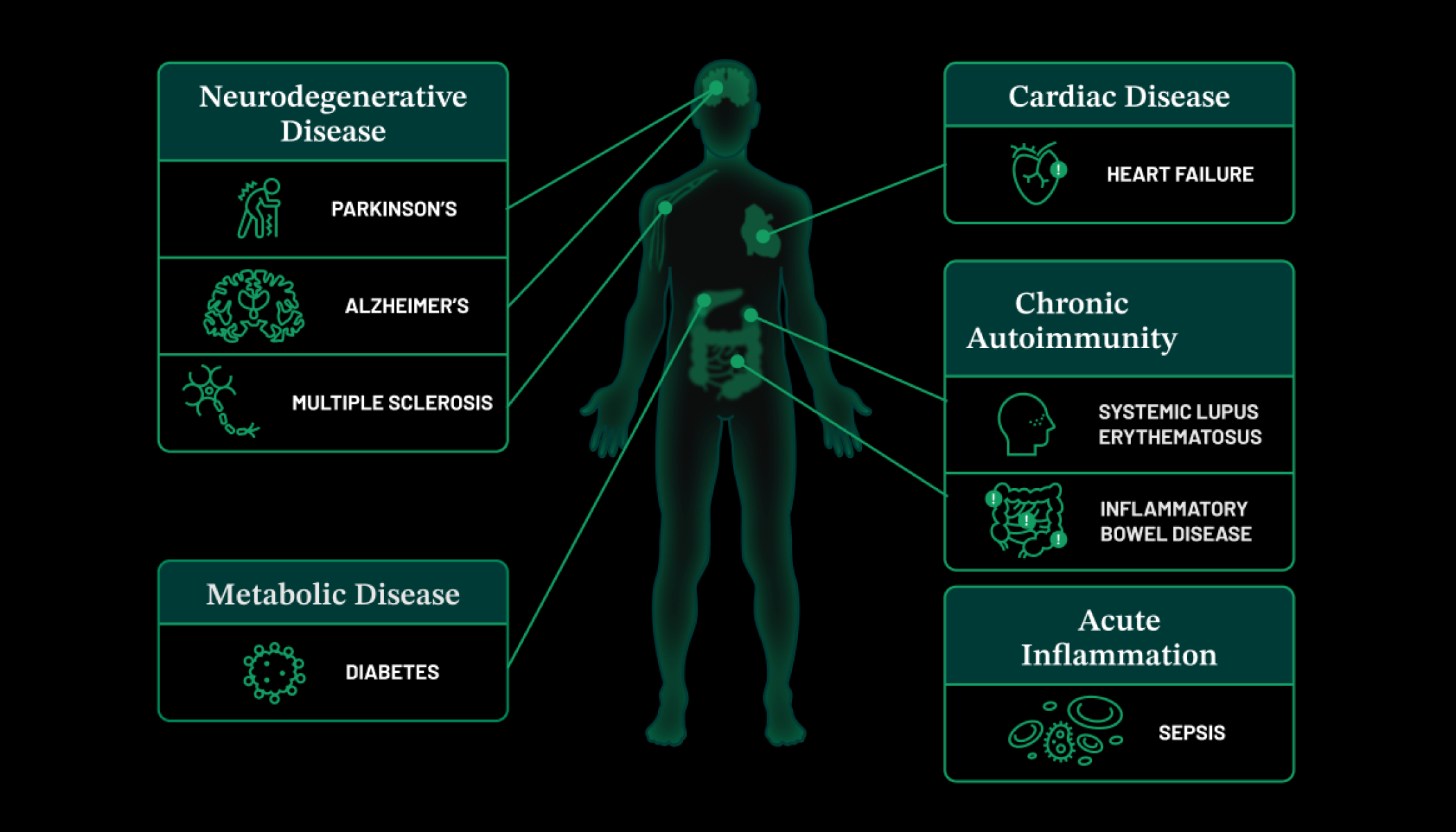
Earlier this month, Mark and I made a big announcement: our next Biohub location will be in Chicago, Illinois, where leading research institutions will build new technologies so scientists can measure biological processes in human tissues, with an ultimate goal of understanding and treating the inflammatory states that underlie many diseases. The Chicago Biohub catalyzes collaboration between researchers from the University of Chicago, Northwestern University, and the University of Illinois Urbana-Champaign to study inflammation. Inflammation plays a key role in many diseases, including cancer, heart disease, and Alzheimer’s disease, and over time, we hope their research will help us better understand these diseases.
This new institute is part of the Chan Zuckerberg Biohub Network (CZ Biohub Network). It builds on a model for scientific and technical innovation that we developed at the first Biohub in San Francisco, California. The key idea behind the Biohub Network is that there are some challenges in science that simply cannot be solved in a traditional research environment.
To make transformational discoveries, you instead have to create a space where scientists, engineers, doctors, physicists and more — people who aren’t normally incentivized to work together — can focus on a wildly ambitious scientific challenge that requires technological innovation and a 10- to 15-year time horizon to solve.
The work is not without risk and there are no guarantees, but it’s the type of work that can also yield massive breakthroughs in how we understand and treat diseases. To share just one example, experts from Stanford University and the San Francisco Biohub are developing a promising new COVID-19 vaccine that’s designed to protect against all known variants, is inexpensive to produce and can be stored at room temperature, which could make Covid vaccines more accessible to people around the world.
I can’t wait to see what the teams in Chicago will achieve in the years ahead.
A Tool To Map Every Cell in the Human Body

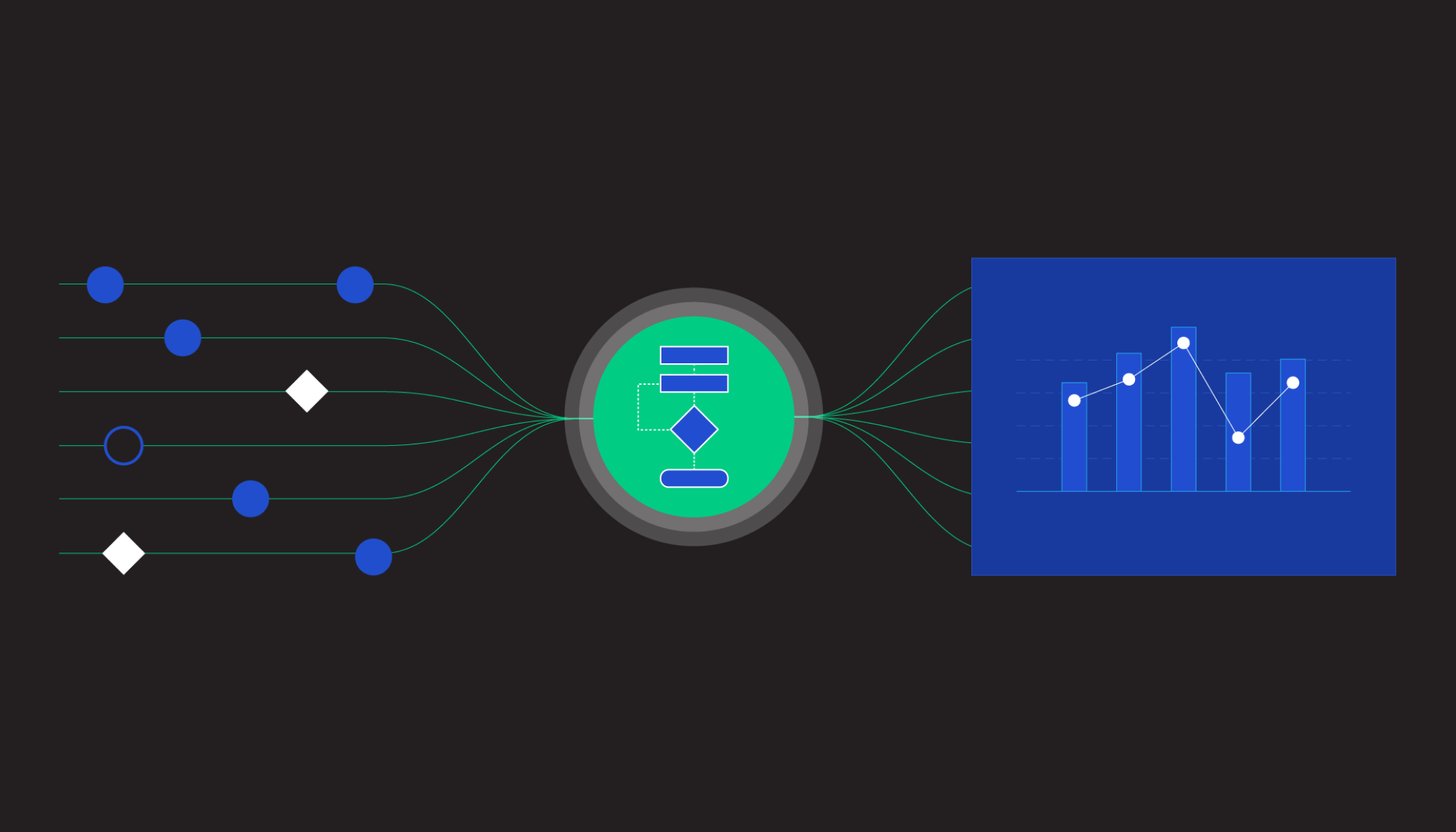
Building technology is one thing that sets CZI apart from other philanthropies. We work closely with researchers to identify the barriers that stand in the way of discovery. Then we build tools to help remove those barriers.
One tool we’ve built is called Chan Zuckerberg CELL by GENE (CZ CELLxGENE). It’s a free, open source software platform that helps scientists share, explore, visualize and analyze data from nearly 40 million human cells.
Thousands of researchers use CELLxGENE every week. Many of them use the tool to rapidly glean insights from large collaborative, global research projects like the Tabula Sapiens and the Human Cell Atlas — which both aim to create an open, shareable reference map of every type of cell in the human body. You can think of it like the Human Genome Project, but for cells.
Researchers are already using these cell atlases to make important discoveries — and even save lives. Before we knew much about SARS-CoV-2, one group of scientists used Human Cell Atlas data to pinpoint cell types in the nose as key players in the transmission of the virus. Their research was an important indicator that wearing masks could help us stop the spread of COVID-19. This work was also a reminder that knowledge of how cells vary across the body can help us rapidly understand diseases, even those that we didn’t know existed only a few months prior.
That’s just the tip of the iceberg. Go behind the scenes of CELLxGENE to see how cell atlases are already transforming our understanding of health and disease.
Pushing for Groundbreaking Treatments for the World’s Rarest Diseases
One of the reasons we build new tools like CELLxGENE is so they can accelerate basic science on a broad scale. Researchers can use CELLxGENE to ask questions about both common and rare diseases, which is especially important for the more than 300 million people worldwide, including children, who have rare diseases.
I met some of these children when I worked at a national referral center for rare pediatric diseases. I also got to know their families, who are some of the strongest people I’ve ever encountered. Many of them are working night and day to bring rare disease patients together, advocate for research, and push for cures.
CZI is proud to fund these organizations through our Rare As One Network. In nearly four years, our partners have brought 600 researchers into collaborations, established 24 patient registries and 19 biobanks, supported 135 research projects, and facilitated the launches of 17 clinical trials.
Behind every one of those statistics, there are stories of hope — and progress — in the face of impossible odds.

For 10 years, doctors couldn’t explain why Kim Nye’s daughter, Tessa, was having over 100 seizures a day. After Kim gave birth to her son, Colton, he started having seizures, too. She started talking to scientists, who eventually diagnosed her children with a form of epilepsy called SCL13A5 Citrate Transporter Disorder — the first known cases in the world.
Kim launched the TESS Research Foundation to advocate for research around the disease. That led to a study that documented how the disease unfolds over time, and soon after, clinical trials for a promising gene therapy. The whole process — from identifying the rare disease to testing the potential treatment — occurred in just four years. Learn more about Kim’s work in our latest video blog featuring rare disease patients and their families who are accelerating research to find treatments and cures.
For me, this is what it means to build, fund and do awesome science — and to build a better future for everyone.
If you want to follow along with our work in science, you can sign up for updates. Stay tuned for my future blogs, which will spotlight our work to support students, educators and our Bay Area community.
With care,
Co-Founder and Co-CEO
Chan Zuckerberg Initiative