Oct 11, 2023 · 10 min read
Marking the Milestone of the Human Breast Cell Atlas
The world’s most comprehensive single-cell atlas of healthy breast tissue furthers our understanding of mammary biology and progresses treatments for diseases like breast cancer.
The Human Breast Cell Atlas, recently published in Nature, is a landmark achievement for mammary biology as the world’s most comprehensive atlas of healthy breast tissue.
The current “atlas era” of cell biology represents a marked shift in the field. Whereas the physiological importance of the cell was not realized until nearly two centuries after their initial 1665 discovery, reference atlases have unraveled a new layer of information by cataloging the molecular landscape of single human cells across ages and dynamic states. This new layer of information is setting the standard for how we define and study cells.
For example, the CZ Biohub’s Tabula Sapiens is a human reference atlas that sampled multiple tissues from individual human donors and performed coordinated single-cell transcriptome analyses on live cells. The Tabula Sapiens samples multiple organs from the same individuals to control for interpersonal variability in cell types and states. Comprising nearly 500,000 cells from 24 different tissues and organs, many from the same donor, this multi-organ approach led to the characterization of over 400 unique cell types. Both sampling breadth and depth are crucial to understanding the variability of human health.
The Human Cell Atlas is an international consortium that charts the cell types in the healthy body, across time from development to adulthood, and eventually to old age. Collaborative groups worldwide continue to contribute data from different tissues to the Human Cell Atlas, including the human lung, retina, kidney, heart, reproductive cells, and now, breast tissue. These atlases help us better understand fundamental biology and illuminate novel therapeutic strategies and targets.
This will be a very impactful resource for researchers studying normal mammary development, breast cancer or inflammation like mastitis.
A monumental seven-year project, the Human Breast Cell Atlas (HBCA) is a collaboration between the labs of Dr. Nicholas Navin of the University of Texas MD Anderson Cancer; Drs. Kai Kessenbrock and Devon Lawson of the University of California, Irvine; and Drs. Bora Lim and Alastair Thompson of Baylor College of Medicine. We spoke to the lead principal investigator on the project, Nicholas Navin, about this work. Read on to learn more about the Human Breast Cell Atlas and what’s next for the team.

Key Findings From the Human Breast Cell Atlas
Single-cell projects at the scale of the HBCA provide a high enough resolution to identify rare subpopulations that smaller studies may be unable to uncover. Using single-cell and spatial transcriptomic and proteomic methods, the HBCA profiled 714,331 cells from 126 women and 117,346 nuclei from 20 women, identifying 12 major cell types and 58 biological cell states with canonical and novel markers. Importantly, donors were able to contribute tissue samples representing diverse biological states, including parity, menopause, age, and self-reported ethnicity.
Researchers were surprised to find limited basal cell-state diversity among the epithelial cells they profiled, whereas epithelial cells in the luminal areas comprise 10 cell states with diverse biological functions. Notably, no proliferating cells or cells with stem cell markers were detected in the basal cell region, raising questions about the presence of basal stem cells fuelling epithelial homeostasis.
Most previous studies focused on epithelial cells in breast tissue, given that these typically give rise to cancers. Navin’s group, however, has had a long-standing interest in the microenvironment of breast tissue, including non-epithelial cell types in connective and adipose tissues. With non-epithelial cell types being understudied, the team was driven to develop an atlas to establish a baseline for typical cell function and uncover cell interactions that may lead tissue toward disease.

In the non-epithelial compartments of breast tissue, including adipose and connective regions, they found an unexpectedly abundant (15.6%) and diverse milieu of tissue-resident immune cells congregating in both the lobules and ducts. Notably, B, T and myeloid cells were roughly 12-14% of all cells. This offers a deeper sense of the immune involvement in breast biology and opens up a wider range of investigation of the immune microenvironment in cancer progression and treatment.
“Prior to this, there were a few reports about immune cells in normal breast,” Navin says. “But nobody had systematically shown these numbers and the variation. So that was a pretty big surprise for our project and, I think, for mammary biology in general.”
Multimodality Accelerates Characterization of Cell Types and States
To catalog a diversity of cells of the breast, researchers applied multiple single-cell transcriptomic and spatial methods, including:
- Single-nucleus RNA sequencing (snRNAseq)
- Single-cell RNA sequencing (scRNAseq)
- Single-molecule fluorescence in situ hybridization (smFISH), including Resolve and MERFISH platforms
- Spatial proteomics
- Spatial transcriptomics
This multimodal approach produced a richer dataset, and helped overcome some of the challenges notorious to breast tissue. For example, fatty tissues interfere with probe hybridization for in situ methods, and breast tissue density is non-uniform, with some areas of tissue being incredibly dense and others more sparse.
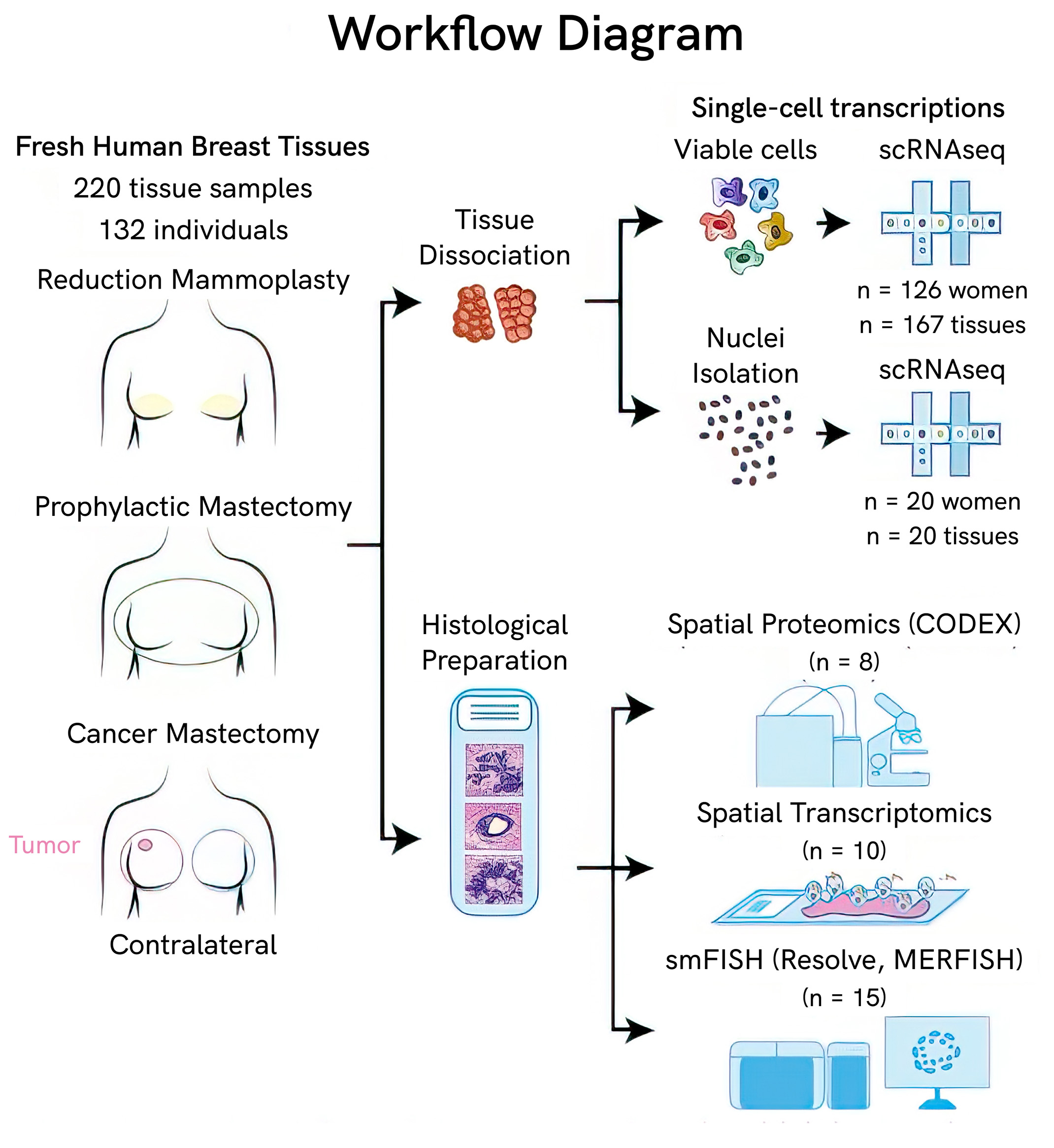
“The different modalities taught us a lot. Initially, we did single-cell RNAseq and noticed we didn’t have any adipocytes, even though we knew those are a big component of breast tissue,” says Navin. “We figured out that those are large cells that are hundreds of microns in diameter, whereas the droplets [in the microfluidic capture system] are 50 microns, so they just don’t enter the droplets.”
That’s where the single-nuclei RNA sequencing came in. While cell size can range drastically, nuclei are relatively uniform across cell types in this tissue, meaning they could get transcriptional data from adipocytes through nuclear capture. This method also revealed other cell types, like mast cells, which were not in the cellular data either.
But the HBCA didn’t stop there.
Navin says, “scRNAseq is the workhorse in many projects, but it’s just the starting point. It tells you what’s there and what different cell markers are. Pretty quickly afterward, you want to know which cells are next to each other, who’s interacting?”
To that end, the collaborators employed various spatial platforms. While they are a newer technology, they enabled the team to see how cells are organized in tissue, account for inconsistencies between RNA and protein levels, and validate selection biases from cell survival after dissociation.
For example, the HBCA found variations in cell states depending on age, self-reported ethnicity and menopause. Presumably, future studies sampling from lactating or developing individuals and further ancestries would uncover additional cell types and states, reflecting the true complexity of human tissues.
Seven Years in the Making
Funded by CZI as part of the Seed Networks for the Human Cell Atlas, the HBCA project took seven years to collect and publish its findings and is a project with many more years of discoveries ahead.
Challenges in Producing a High-Quality Atlas
“People ask why it took seven years,” Navin says. “But a lot of it was the validation and the spatial analysis that we thought was really important.”
According to Navin, the biggest challenge in the project’s early days was standardizing protocols between the two groups at MD Anderson and UC Irvine. They realized the impact this could have on their data and wanted to ensure they consistently integrated the results while minimizing the loss of biological signals.
“We got early data back that said which critical parameters have the biggest influence on skewing the cell types and representing them. And for us, it was the dissociation time: the amount of time that the cells were in the enzymatic buffer,” Navin explains.
For example, they found that leaving tissue in the dissociation buffer for longer enriched some cells, like myoepithelial cells, and led to fewer immune and vascular cells. By systematically testing different incubation periods, they found that three to six hours was the sweet spot for all the cell types they hoped to capture.
Ultimately, the time was necessary to ensure high quality —something Navin and the team did not want to compromise knowing the future uses of such a large reference atlas.
“We knew it was going to be analyzed and referenced a lot, so we did spend quite a lot of time making sure we could provide the best possible dataset,” he says.
Open-Access Cell Atlases Pave the Way for Future Research
A key to the success of this project was collaboration. With all of the protocols and data that went into the HBCA available completely open access, Dr. Navin hopes the collaborations will continue to grow. All of the data is on CZ CELLxGENE, allowing other researchers to search for genes of interest and perform other analyses within a web browser.
“CELLxGENE [Census] also lets other researchers download the raw data and reanalyze it very quickly,” Navin adds. “And maybe people will do that and disagree with us or say something should be labeled differently. But that’s the whole point of putting it out there, so that we can have a discussion and compare data.”
An Atlas Is Just the Beginning
As with most atlases, the work doesn’t end with publication.
While the depth of the data provides numerous exciting next steps, Navin expressed a few areas of particular interest to their collaboration.
First, “continuing to build this atlas and integrate atlases. While not without challenges, that’ll allow us to increase the dataset and also the ethnic and ancestral diversity, across different cities in the United States and the world,” he says.
In fact, earlier this year, a group in the UK shared a single-cell breast atlas created from 55 donors who had undergone reduction mammoplasties or risk reduction mammoplasties.
Navin explains, “Even though we have quite a large dataset right now, we realize that if you want to look at a lot of the rarer cell states and things that are not in majority of the women, you need much more statistical power.”
This is critical for a future of precision health, where everyone is represented so everyone can benefit.
Following up on the various cell types, including immune, stromal and epithelial cells, would be of interest to better resolve their variations and functions.
But for Navin, the comparison to disease states was one of the big motivations for working on the HBCA.
“We’re going to look a lot at premalignant breast cancer, invasive breast cancers and triple-negative breast cancers to understand how they are different from baseline,” he says. Building off the diversity of cell types in the atlas, they can look “not just at the cancer cells, but all the components of the tumor microenvironment, immune cell populations, fibroblasts and vasculature.”
In addition to their work, the team welcomes others to work with and contribute further to the dataset and, subsequently, the single-cell and spatial characterization of mammary biology.
Learn More
Human cell atlases are still in development for organ systems across the body. Learn more about funding opportunities in this area and discover new single-cell atlas data on CELLxGENE.