Jan 25, 2021 · 9 min read
The Past, Present, and Future of Medical Imaging
Imagine you feel shortness of breath and chest pain, and your doctor isn’t immediately sure what’s causing your symptoms. Today, the next step would be to run a slew of tests and hope that an obvious cause presents itself.
But the future of health care may look very different. As medical imaging technology advances, physicians may one day be able to look directly at the cells in your lungs, observe their activity in real time, and tell you not only about the exact pathology of your problem, but the unique treatment it should receive.
That future might sound like it’s straight out of Star Trek, where Dr. McCoy can wave a tricorder over a patient’s body and know exactly what’s wrong. But more than 400 years of scientific advances suggest that this really is the direction we’re headed — and that amazing new biomedical imaging technologies aren’t that far off.
Discover more: Chan Zuckerberg Institute for Advanced Biological Imaging
Understanding the History of Medical Imaging Technology
To understand where we are today with medical imaging and where we’re going, it helps to look back on the progress humanity has made already.
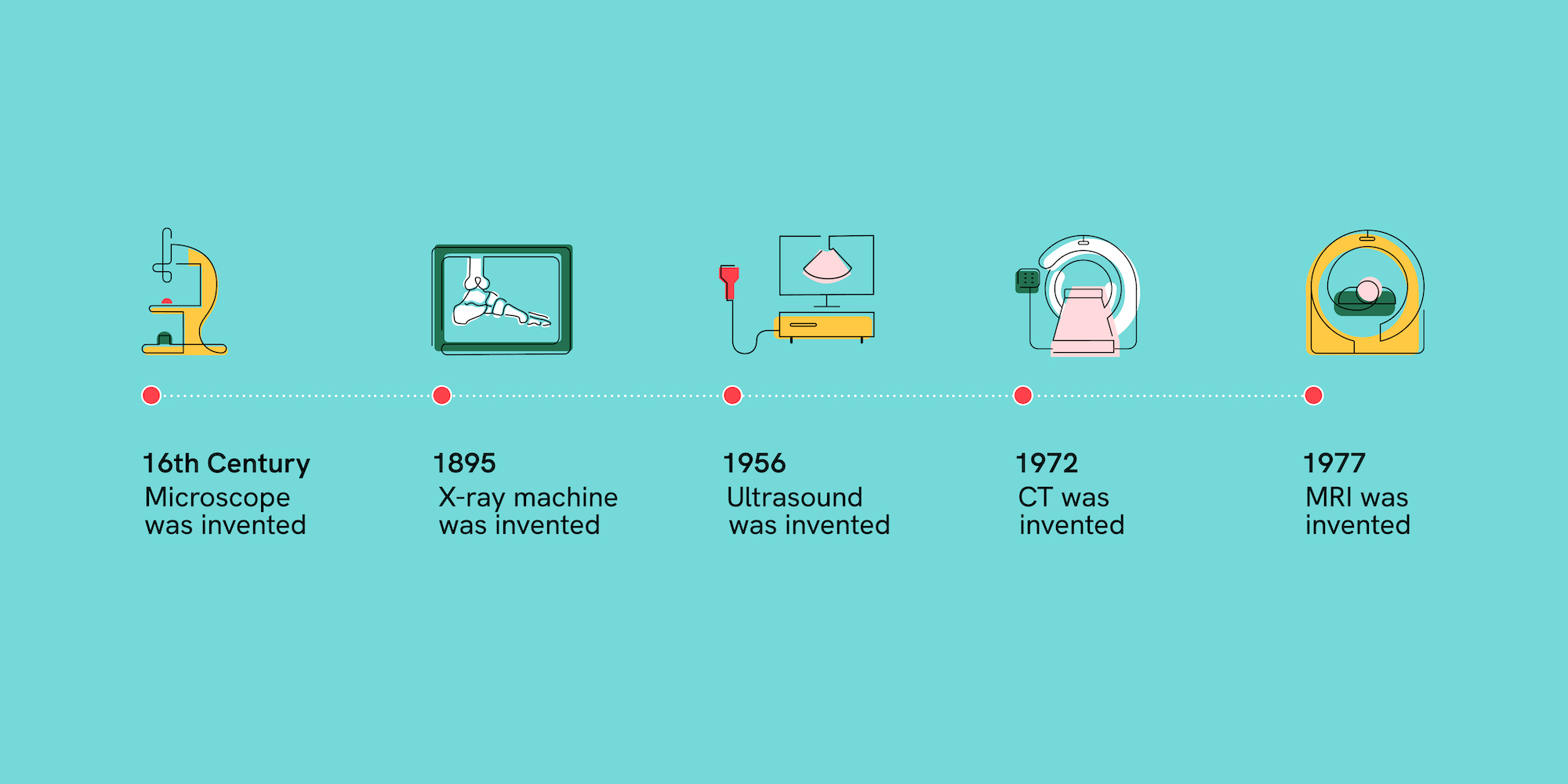
Until the late 1500s, the state-of-the-art imaging technology was the magnifying glass. But then, in the late 16th century, imaging technology made its first great advance with the invention of the compound microscope.
Initially, the compound microscope was mostly used as a source of amusement for curious aristocrats. But then, nearly a century later, improvements in lens technology enabled scientists like Robert Hooke and Marcello Malpighi to start using the microscope to find forms of life previously unseen by the naked eye.
While Hooke used the tool to explore the world around him, eventually identifying plant cells in thin slices of cork, Malpighi looked to better understand the human body: he examined the brain, spleen, and other organs to discover capillaries.
Antony van Leeuwenhoek then took this work to another level. A merchant by trade, he was able to develop lenses to provide new levels of magnification, and used his creations to look at everything he could get his hands on: insects, animal hair, pond water, and even what he scrubbed from his teeth. It was inside that latter sample in 1676 that he discovered small organisms that humans had never previously seen — bacteria.
Of all the scientists who started using microscopy to examine human cells, however, perhaps none had greater impact than Spanish scientist Santiago Ramón y Cajal. Using the staining technique pioneered by Italian scientist Camillo Golgi, in which cells inside an organism are stained with chemicals, Ramón y Cajal was able to determine that the nervous system was made up of individual cells. This discovery in 1888 confirmed that the nervous system is not made up of a single, continuous network of fibers — as was popularly believed — but instead a vast collection of distinct, individual cells, a finding that became the basis of modern neuroscience.
By the close of the 19th century, entire worlds, including those inside of us that had long been invisible, became visible. But how to use the knowledge of these worlds to improve our health remained a mystery.
Rapid Advances in Medical Imaging Technology During the 20th Century
As useful as existing imaging technologies were in exploring the composition of the human body, there were still major obstacles that prevented them from improving human health. This was largely because for most of the 19th century, getting a close look at a sick person’s cells required all kinds of impractical — and invasive — procedures, including surgery, tissue removal, tissue staining, and more.
That began to change with Professor Wilhelm Röntgen’s invention of the X-ray in 1895. Scientists gained the ability to see inside live patients, instead of only their lifeless tissues, and visualize the inside of the human body without cutting it up into tiny pieces.
It’s no wonder that within a year of Röntgen’s announcement, X-ray machines became a key part of the medical profession — and in so doing, kicked off a century of progress. One advance in imaging technology led to the next, and barriers that had long limited better diagnostics and treatments continued to fall.
And when X-ray technology couldn’t tell us much about soft tissues in the body, early ultrasound technologies — which built upon the use of sonar during World War II — were developed to do just that. Not long afterward, the advent of tomography techniques beginning in the 1960s — like CT and PET scans — made it possible to build 3D images.
It’s hard to overstate how far the field of medical imaging advanced in a single century — from Ramón y Cajal’s neurons on a slide to a three-dimensional image of a living brain as seen through an MRI scan. These technologies provided new insights, giving medical professionals crucial added context to diagnose and treat patients more quickly.
Also read: 5 Scientists Pushing the Boundaries of Imaging Technology
How Medical Imaging Has Revolutionized Medicine
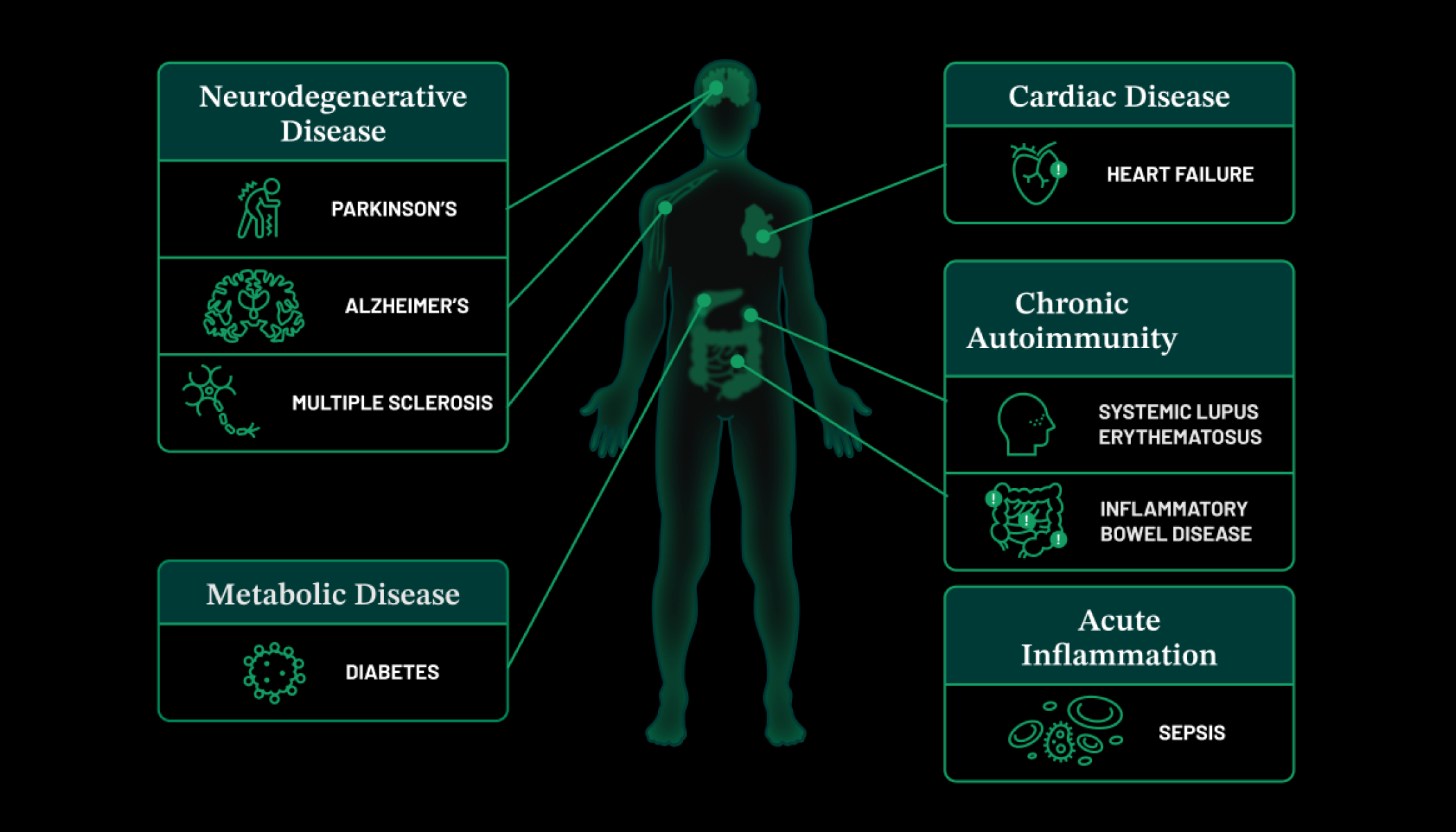
But of course, there are still more frontiers ahead — and more opportunities to use imaging to revolutionize medicine. As living, breathing beings, our biological systems are in constant communication: tissues, cells, and proteins interacting with each other, and with other parts of the body. Without visibility into all of these processes, we’re only scratching the surface in our understanding of the human body, and how to protect it from disease.
Take cancer, for example. With the technologies we have now, we can visualize tumors inside the body, but we cannot see them at the cellular level of depth and resolution that we need. We cannot see the tumors grow in real-time, or watch the cancer cells interact with drugs or immune cells, or see any cancer cells that may remain after a tumor is extracted. If we could, we would understand and treat cancer much more effectively.
To get to that new level of understanding, however, we have a number of challenges to solve as a scientific community.
First, we need to be able to see deep inside the body at a high resolution. Right now, MRIs, sonograms, and other techniques can noninvasively provide images from deep in our bodies. But the resolution is too low to actually see cells interacting.
We do have other technologies that achieve high levels of resolution, but these have the opposite problem. Electron microscopy, fluorescence microscopy, and other technologies allow us to see high-resolution images of tissues and cells that are outside the body. But these techniques can’t show what’s happening between and within the cells that are actually deep inside our bodies.
The Role of Deep Tissue Imaging
And this is where deep tissue imaging comes in. It’s a promising set of to-be-developed technologies that could allow researchers to observe cells at high resolution through skin and bone, watch cells divide within tumors, and track how the body responds to drugs in real time.
But that’s also where a second challenge arises. Because deep tissue imaging collects and interprets multiple sources of information, from sonograms to CT scans — and does so not just for a moment, but over time — it presents enormous data processing challenges. Right now, we don’t have the technology we would need to interpret those data in an accessible, scalable way. If we want to actually unlock the potential of this technology and put it in doctors’ hands, we’ll also need to develop new ways to organize, process, and store the data it generates.
In a way, we find ourselves at the same place where scientists throughout history have arrived: a barrier (or two) away from a breakthrough. And on the brink of seeing farther than we ever thought possible.
Looking Toward the Future of Medical Imaging Technology
Yet, for all we can’t see, there’s one thing that’s immediately clear: the imaging scientists that we’re working with at CZI are leading the way in solving the imaging challenges we face.
Take Dr. Mikhail Shapiro of the California Institute of Technology, who’s pioneering a new deep tissue imaging technique to observe cellular functions deep within the body. He’s engineering cells to form air-filled proteins, which cause the cells to stand out in ultrasound images and allows them to be more easily tracked over time — a process he’s already using to investigate the behavior of cancer cells.
Or consider Dr. Laura Waller, a CZI grantee using computational microscopy to image previously unattainable information. By using new techniques, such as 3D phase microscopy, she is providing us new views into the deepest structure of organs like the brain — a crucial step to understanding neural activity.
Also read: A Day in the Life of an Imaging Scientist: Laura Waller
In the near future, these tools could help us develop personalized care for our illnesses. For example, a cancer patient may not need to undergo a blunt force approach like chemotherapy after their diagnosis. Instead, their doctor may target specific cells based on their genetics, or particular proteins gone awry, to fight the disease.
Further on the horizon, we can imagine more revolutionary changes to how the medical system works. That same doctor may be able to identify risk factors for their patient even before the cancer cells appear, and take early steps to prevent the illness from ever taking hold.
As Dr. Shapiro has said, “to look is the most basic human instinct.” For hundreds of years, imaging scientists have been looking ahead and bringing us closer to imagining a world free from disease. Their work reminds us that if we go deep and then deeper still, anything is possible.
This article was last updated on July 25, 2022.