Jul 27, 2023 · 9 min read
10 Questions Revealing the Mysteries of Human Cells
We answer frequently asked questions about the microscopic cells that make up our bodies.

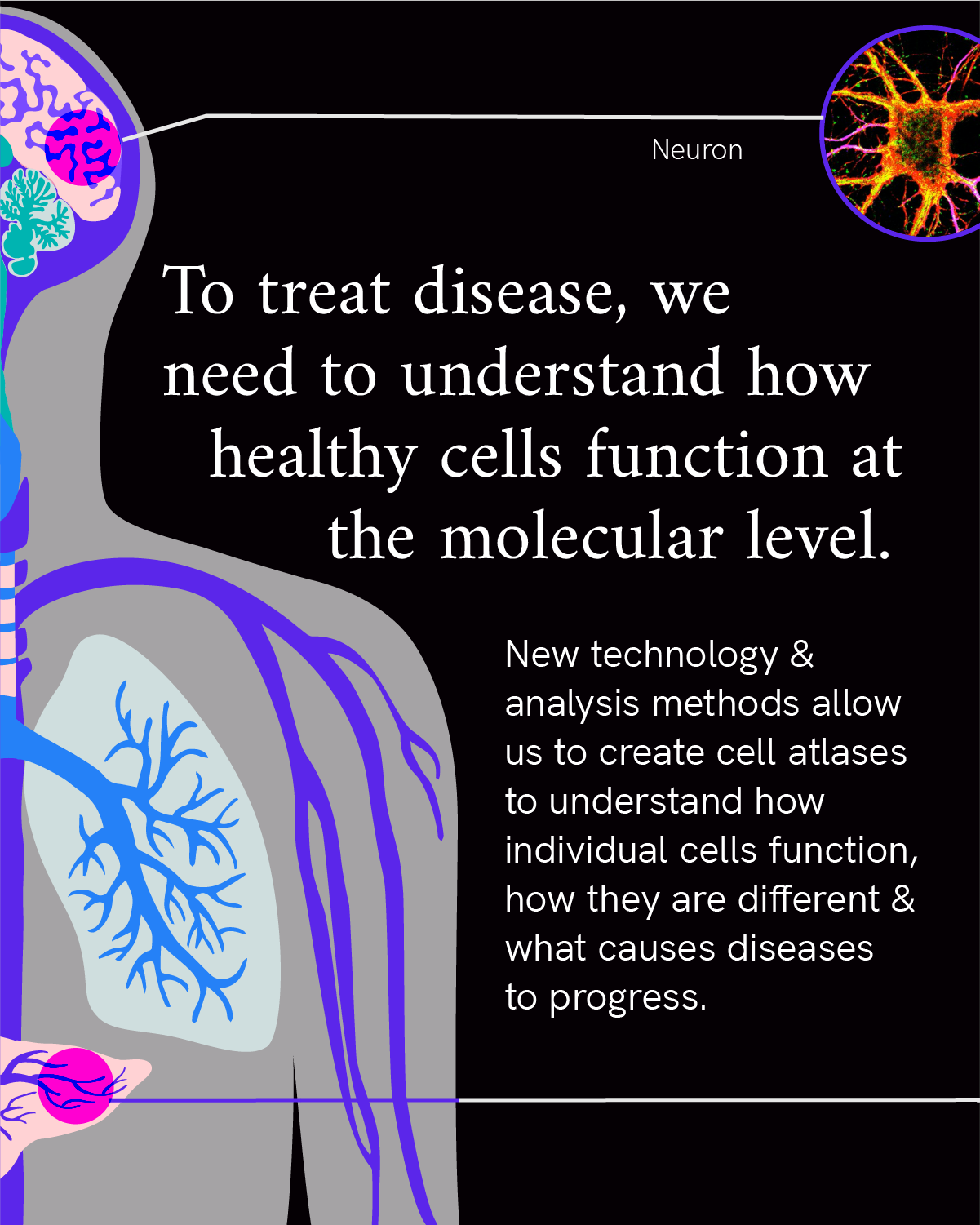
Cells, smaller than the eye can see, are the fundamental unit of our biology. From the very first cell of our body — the fertilized egg — all the cells of our body are made and formed into the tissues and organs that help us breathe, think, move … be. The cell is the most fundamental unit of human life. Yet despite decades of scientific research and progress, much of it remains a mystery.
That’s why over the next 10 years, CZI aims to understand the mysteries of the cell and how cells interact within tissues and organs. By understanding biology at the microscopic level of our cells, researchers can unlock new insights into how our bodies function in health and the changes that underlie disease. Focusing on fundamental science to better understand human biology will lead to groundbreaking treatments and therapies.
Keep reading for some frequently asked questions about cells and learnings from the latest research.
1. How many cells are there in the human body?
The current best estimate is that the average body contains about 37.2 trillion human cells. There are more cells making up the human body than the number of seconds in one million years. And about 372 times as many cells making just one body than there are stars in our home galaxy, the Milky Way. And that’s without factoring in the trillions of single-celled microbes living throughout the human body in symbiosis.
These trillions of cells are the basic functional unit of our biology, coming together to form the different organ systems in our body. Cells come in many forms and functions — from elongated cardiomyocytes that help the heart contract, to the tree-shaped neurons that transmit electrochemical messages throughout the brain, and biconcave, disc-shaped cells carrying iron throughout our blood. Researchers are working to map all of these different cells in our body to better understand health and disease.
2. How big is a cell?
Human cells come in a range of sizes, though most are too small to be seen with the naked eye. The main exception to that is the ovum, or egg cell, which is among the largest cells in the body at about 0.1 millimeters (or 100 micrometers) across. Rival to the unusually large size of the ovum are motor neurons emanating from the spinal cord down to the biggest toe. These are about 100 times thinner in diameter than the ovum, but have a single projection running the length of the leg, reaching up to one meter long.
Most other human cells average about 10-100 micrometers in diameter. To picture that scale, imagine a single grain of salt cut into five pieces. Each of those pieces would be about the size of the average human cell — no longer visible without magnification, and so small that about 635 of these cells could fit across the diameter of a penny.
The size and shape of a cell is closely tied to its function, and can change over time. The growth and division of cells are very carefully regulated to maintain a healthy state.
Read more: #ImagingTheFuture of Biomedicine
3. What are the features of a human cell?
Human cells have a fat-lined membrane on the outside with many proteins embedded in it that can act as sensors for the environment outside the cell.
The inside of the cell is like an underwater city: instead of buildings there are smaller structures called organelles, and millions of protein molecules, both tethered and floating, swimming throughout the fluid inside the cell.
The city center of the cell is walled off by another fatty membrane, shielding the nucleus within from the rest of the activity in the cell. Inside that nucleus, the cell’s genetic instructions coil and uncoil as needed to direct the orchestra of activity in the cell.
Explore more: Biohub OpenCell project
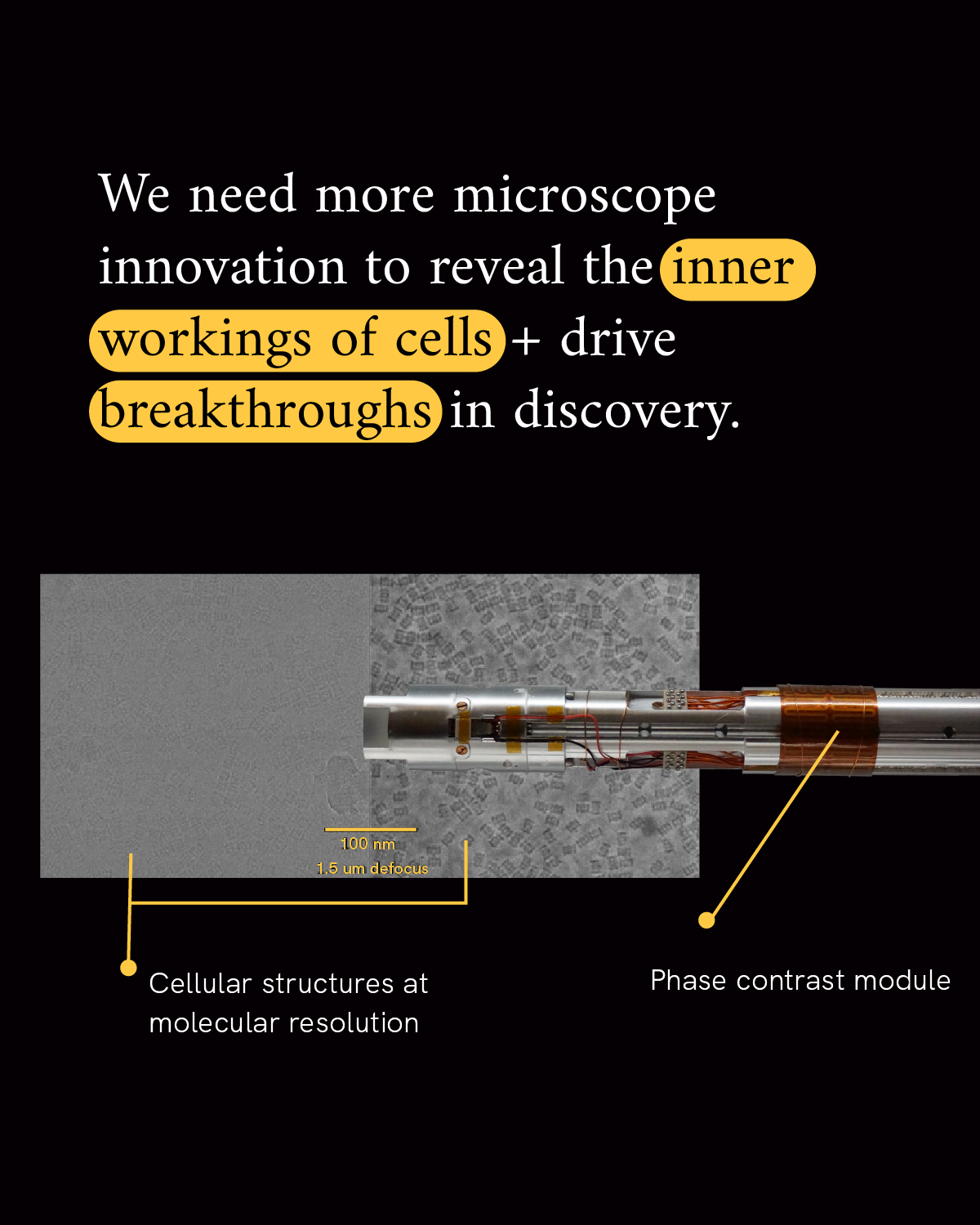
It’s that activity within the cell that keeps our bodies functioning every day. Any changes to that activity are what underlie disease. Imaging scientists are building new microscopes and data analysis tools, plus strategies for sharing them collaboratively, to give us a front row seat to the hum of activity in our cells.
Many of the key active players they wish to observe are many orders of magnitude smaller than our cells. In fact, proteins within our cells are so small that even studying complexes of many proteins combined together requires imaging at the nanometer level. Measuring in at one-billionth of a meter, a nanometer is on the same scale as that same grain of salt chopped into 100,000 pieces.
It takes specialized modern microscopes to resolve the inner details of a cell at the nanometer level. And advanced technological tools and software to analyze the volume of data generated by that level of resolution. For example, imaging just one cell at nanometer resolution takes up the same amount of data as about 83,000 photos on your phone.
The goal of the Chan Zuckerberg Institute for Advanced Biological Imaging is to solve the challenges necessary to increase image resolution and contrast and develop a dynamic, integrated view of biological systems in health and disease.
4. Does every single cell in my body have the same DNA?
For the most part, yes, every cell in the body has roughly the same set of DNA. That’s because all cells in the body originate from the fertilized egg, created through many, many rounds of cell division. Each time the cell divides, it makes a copy of its genetic material so there is enough to be passed on to the new cell.
While there are many “proofreading” steps to reduce errors in the process of duplicating DNA, there are approximately 120,000 copying mistakes across the 6 billion bases in the genetic code every time a cell divides. While most of these changes do not amount to any significant changes in the genetic code, they can accumulate as cells in the body continue to divide over our lifetime.
While every cell in a person’s body has nearly the same set of DNA, cells become different depending on the subset of the DNA being used. It’s similar to how an orchestra works: all of the instruments are there, but not all of them are getting played at once, and depending on the timing and combination of instruments you can get completely different music from the same set of instruments. While every cell has the full set of DNA, as a cell develops and specializes it uncoils the parts of DNA it needs to use and coils up the parts that are less relevant to its functions. That’s how a muscle cell ends up different from a skin cell, even though they have the same set of genetic instructions.
Researchers want to learn more about how differences in people’s DNA, environments, and lifestyles impact their health to formulate more precise treatments and prevention strategies. CZI has partnered with the US’ four historically Black medical colleges to further support their cutting-edge scientific research in this field to accelerate precision health for all.
5. How many different types of cells are there in the human body?
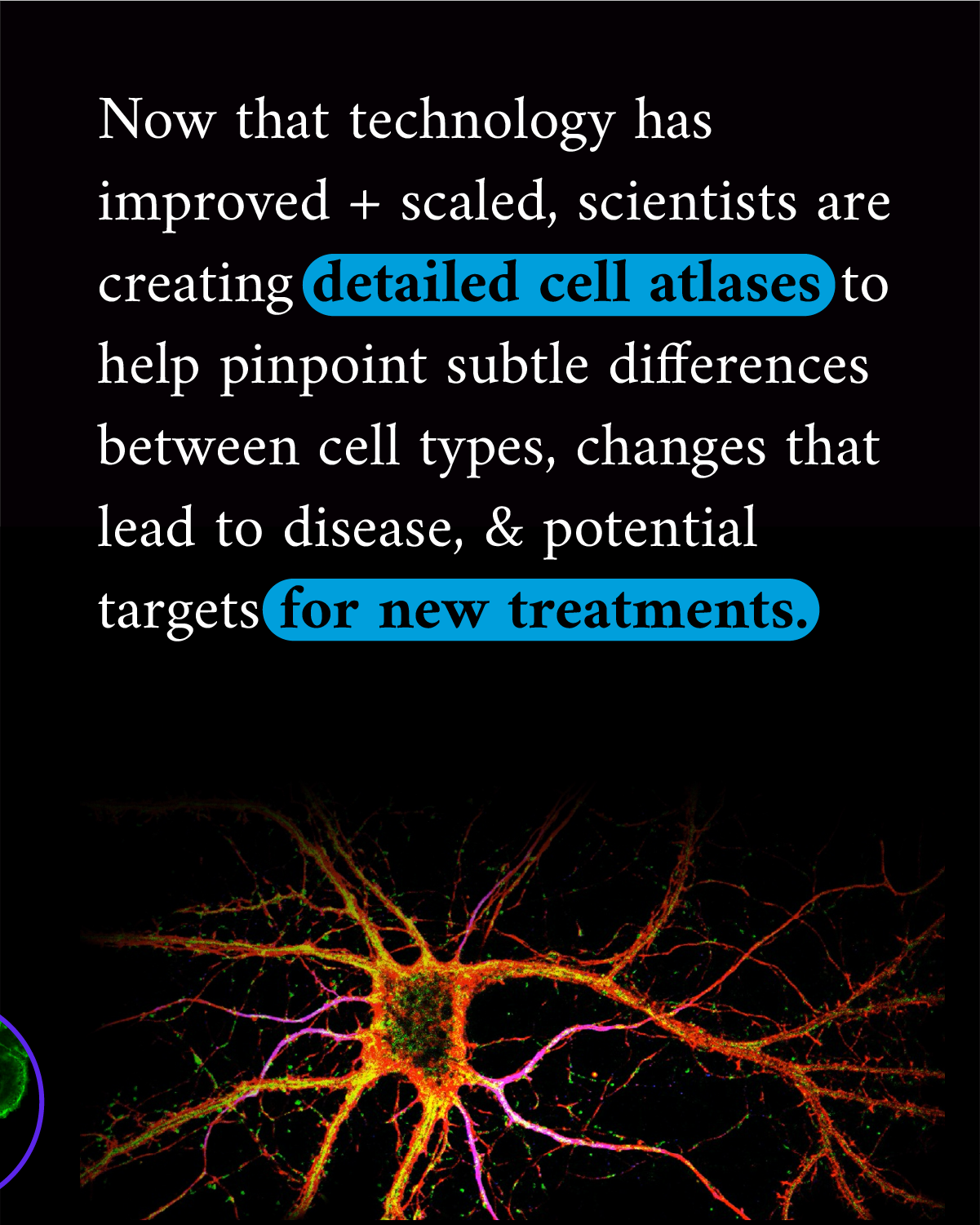
Historically the answer has been that there are about 200 different types of cells in the human body, but new technology in the last decade has uncovered many more than that, totalling a couple thousand.
For example, we’ve known for over a century that there are four main types of cells in the brain: message-conducting neurons, star-shaped support cells called astrocytes, cells that insulate neuronal connections called oligodendrocytes, and specialized immune cells that survey and respond to changes in the brain called microglia. From the first glimpses of these cells through the earliest of microscopes it became clear there are many subcategories of each of these cell types.
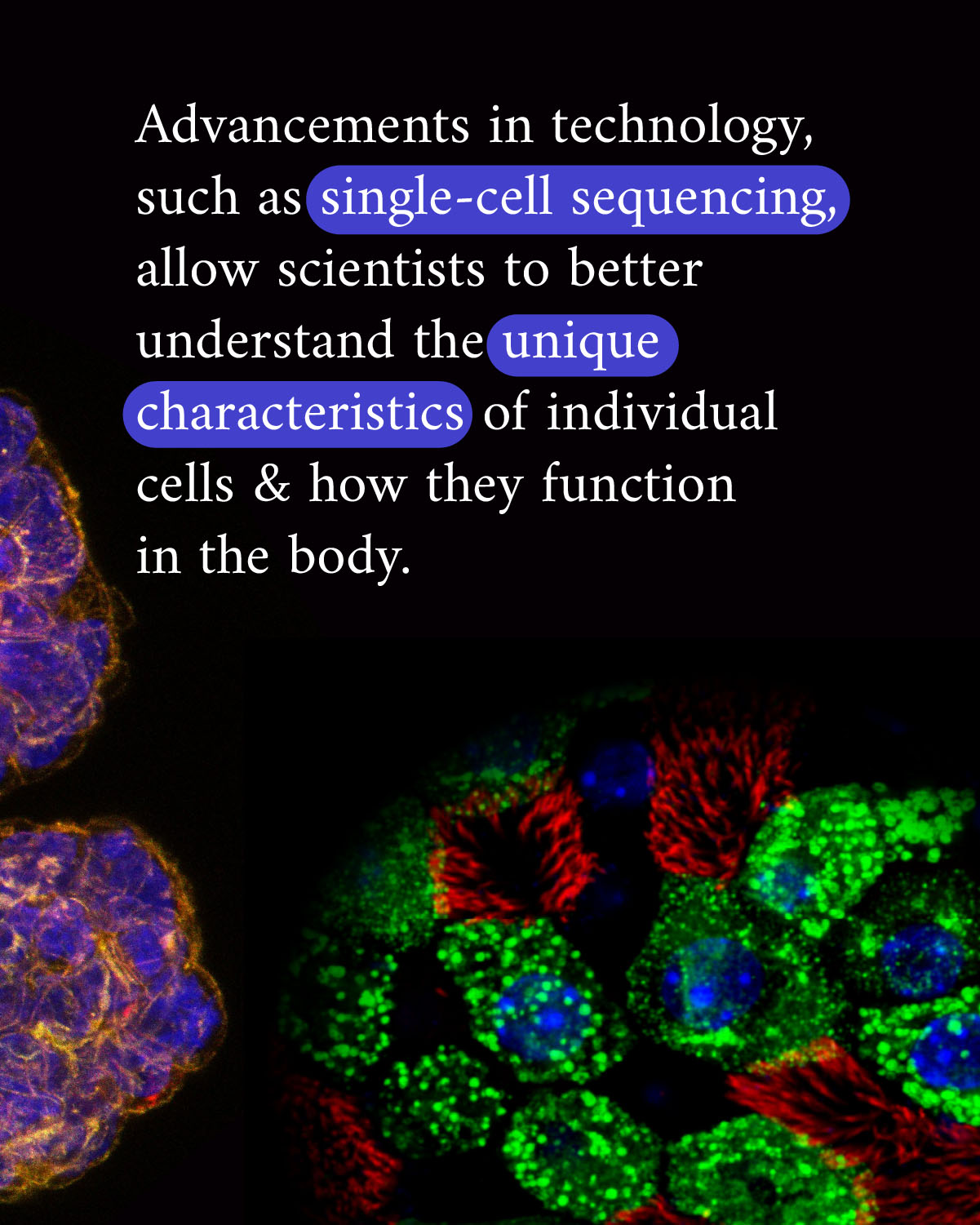
But it’s taken advances of imaging technology and molecular biology to fully begin to realize just how varied cells can be. For example, the Tabula Sapiens is a project by the Chan Zuckerberg Biohub San Francisco and CZI to map cells of the human body using molecular data. They have already characterized over 400 different cell types using molecular data from studying 500,000 cells from 24 different tissues and organs. The data in these comprehensive cell atlases put a spotlight on the subtle differences between cells that are key to maintaining health, and help us better detect the early changes that lead to disease.
6. How do scientists harness all the information of our cells?
New technologies that resolve cells at the nanoscale or map the entire body at single-cell resolution generate a massive amount of data. In the latter case, tools like Chan Zuckerberg CELL by GENE aggregate and standardize data, making it easier to access and analyze. This collaboration of biomedical sciences and software engineering is critical to ensure data can be transformed to understanding the mysteries of the cell. Importantly, building and supporting tools and resources that share values of open science are key to accelerating science around the world.
One of the most important factors to consider when building these maps, however, is that they accurately reflect the diversity across the human population. The Ancestry Networks for the Human Cell Atlas projects support researchers in creating single-cell datasets of tissue samples from ancestrally diverse donors, with the aim of creating a more globally representative resource to understand health and disease.
7. Can cells replace themselves?
Some cells in the small intestine replace themselves every few days, while those in your blood regenerate about every two months. Other cells, like oocytes and those in parts of the brain and eye, do not regenerate at all.
In neurodegenerative diseases like Parkinson’s and ALS, these non-regenerating cells are lost, leading to symptoms of disease. By bringing together researchers from unique disciplines, we want to enable earlier and more accurate diagnosis of neurodegenerative disease and find new treatments.
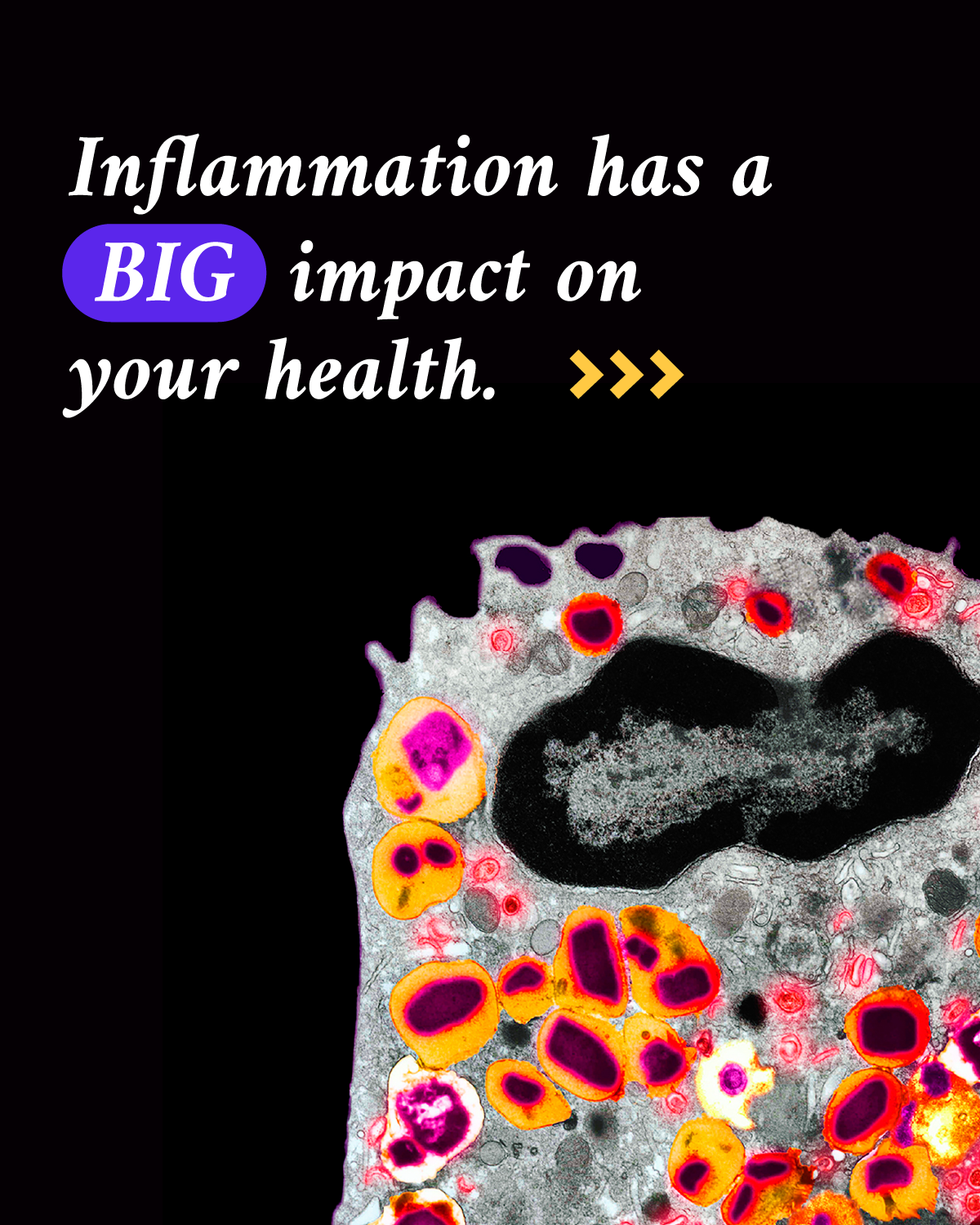
8. What happens to our cells and tissues when inflammation goes into overdrive?
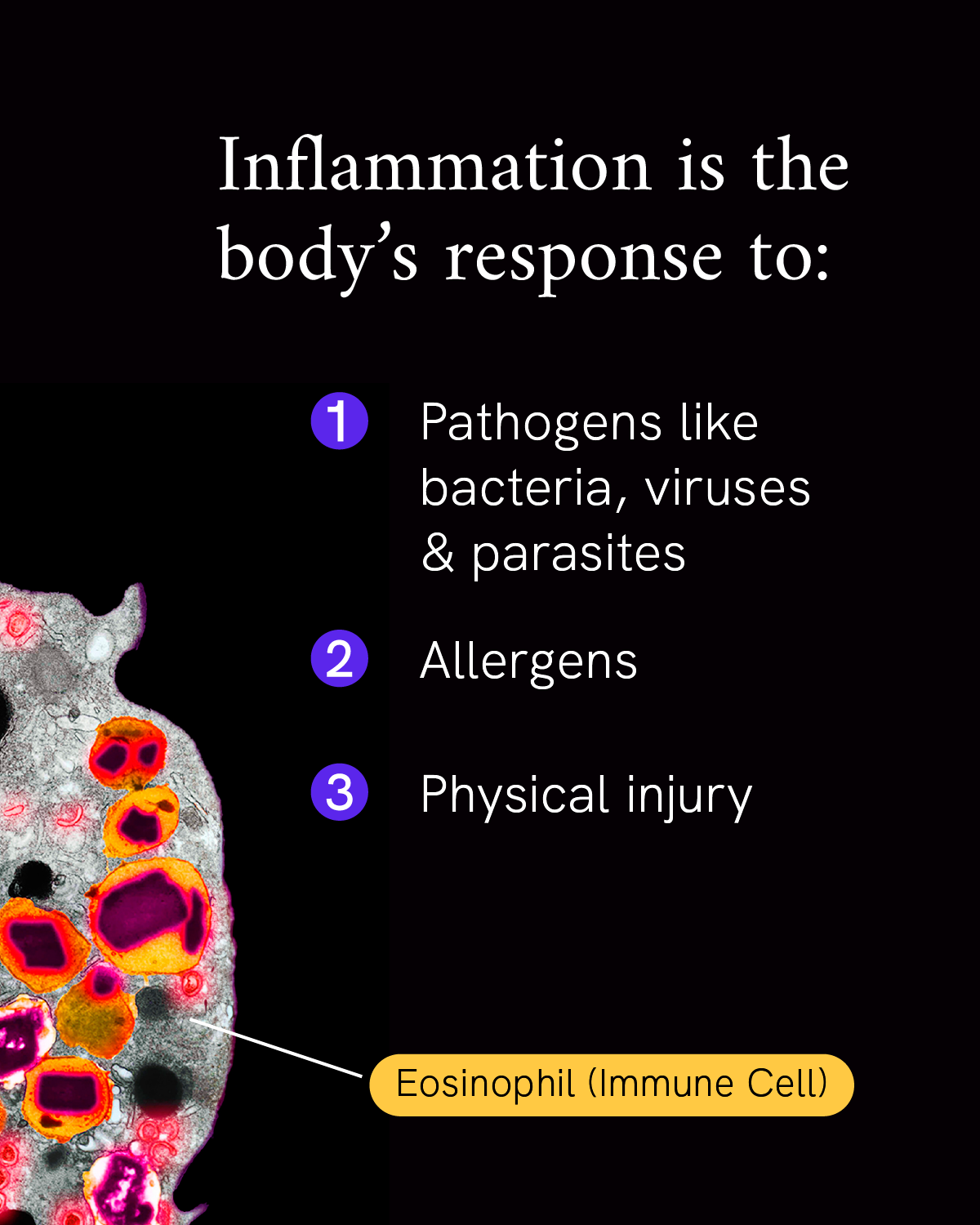
Inflammation is our body’s first line of defense against injury or infection by harmful viruses, bacteria or other pathogens. It’s an important response from the immune system to try to minimize injury or infection and begin the healing process, but too much inflammation at the wrong place or time can become a problem.
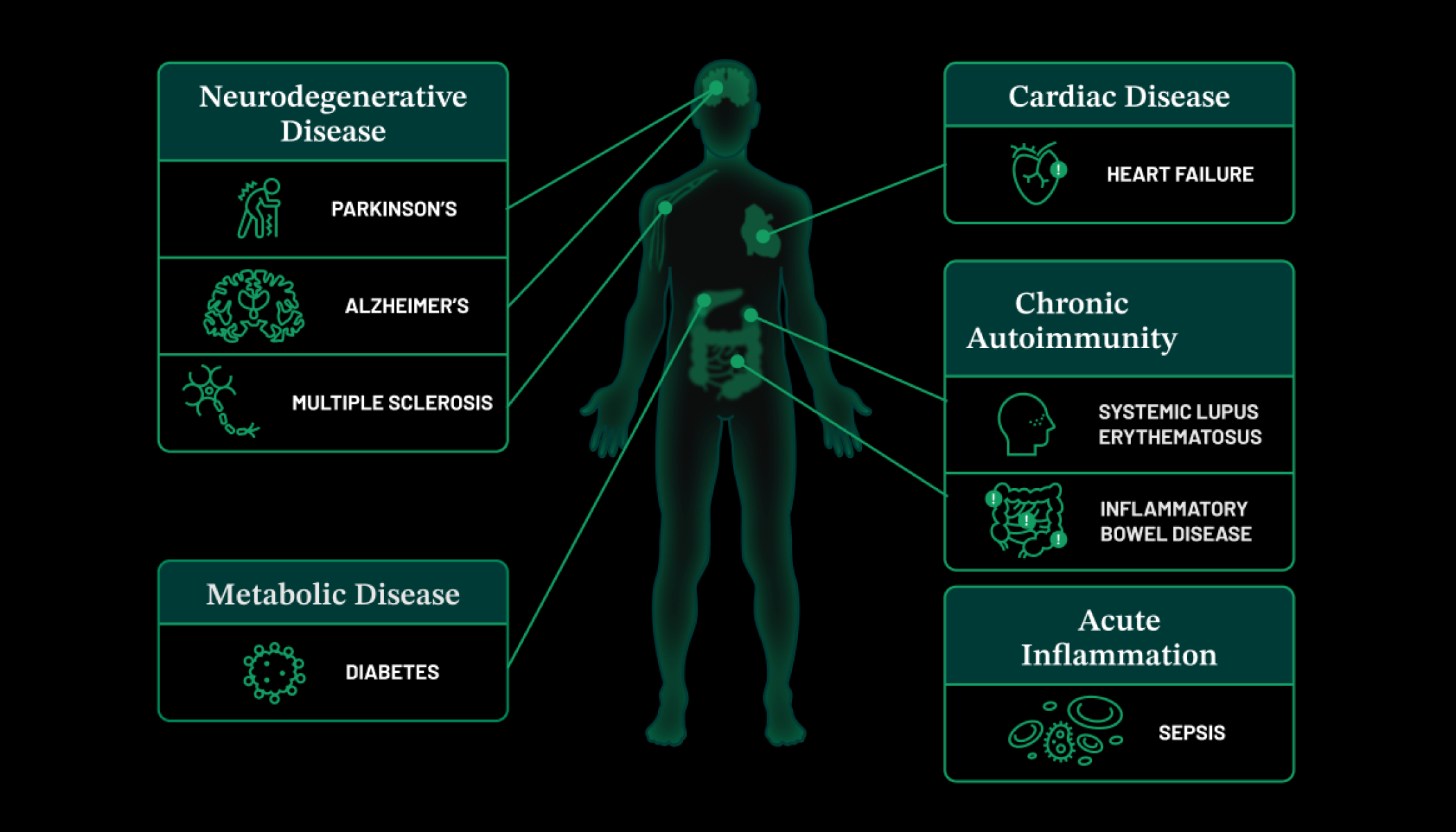
About 50% of all deaths can be attributed to inflammation-related diseases — including cancer, heart disease and dementia. If scientists can watch the human immune system in action to see how our cells and tissues are impacted, they can figure out ways to control inflammation.
Researchers at the Chan Zuckerberg Biohub Chicago are engineering new tools to measure inflammation in living tissues. This will enable them to study the fundamental causes of inflammation — how it starts, why it keeps going, and how to turn it around so that we can prevent disease before it starts, or find ways to treat it at an earlier stage.
9. How does understanding the cell’s response to disease allow us to develop new therapies?
Diseases result from a change in our body’s function, right down to the level of our cells. By studying which cells are affected in a disease and what changes are happening to everyday cell processes, researchers will be better poised to treat the root cause of a disease. That’s because understanding more about the biology of a disease can enable researchers to identify new targets for medications, better understand disease progression, make more personalized predictions of how someone may respond to a treatment, and develop preventative measures for disease.
For example, rare diseases often involve specific genetic mutations or changes to how genes and proteins are regulated, which point to the role they play in our cells. Through the CZI Patient-Partnered Collaborations for Rare Neurodegenerative Disease and Single-Cell Analysis of Rare Inflammatory Pediatric Disease, researchers and patient organizations are partnering to accelerate our understanding of some of the more than 7,000 rare diseases affecting more than 300 million people worldwide. This work not only advances us towards new treatments for these diseases, but gives us a better understanding of basic cellular processes that are important to treating common diseases, too.
10. What are some unsolved problems in cell biology?
Each of the 37.2 trillion cells in the human body is like a whole city of biological activity. Each cell is complex with many parts interacting within an ever-changing environment. There is so much we still don’t know about how individual cells in our bodies change over time and how they interact as systems in our tissues and organs.
While new technology is giving researchers a front-row seat to that activity, collaboration and open sharing of data and tools is key to transforming this new information into scientific breakthroughs.
We are building open source software tools to accelerate science, funding important research and launching institutes to do research that can’t be done in conventional environments.
For the next 10 years, our goal is to understand the biggest biological mystery about the human body — the cell. A foundational understanding of how they work will lead to discoveries that will change medicine in the decades that follow.